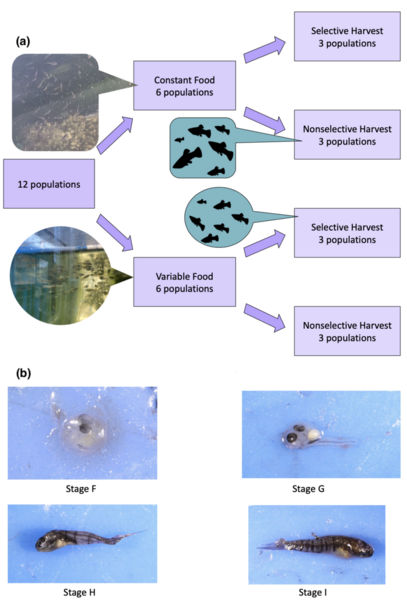
The authors looked at the impact of different harvest and feeding treatments on Heterandria formosa over three generations as a model for changes in marine ecosystems.
Read More...Observing food and density effects on the reproductive strategies of Heterandria formosa

The authors looked at the impact of different harvest and feeding treatments on Heterandria formosa over three generations as a model for changes in marine ecosystems.
Read More...Computational Structure-Activity Relationship (SAR) of Berberine Analogs in Double-Stranded and G-Quadruplex DNA Binding Reveals Both Position and Target Dependence
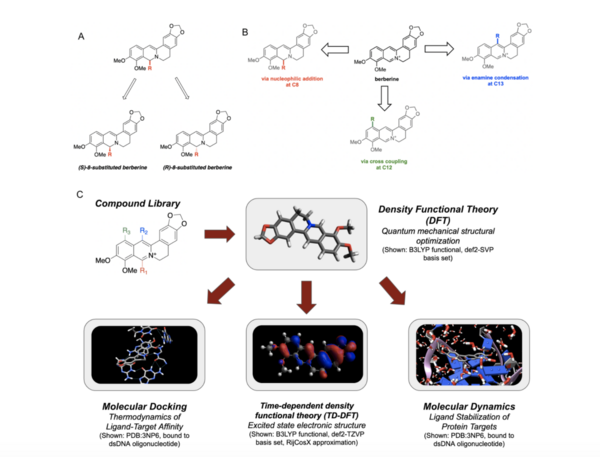
Berberine, a natural product alkaloid, and its analogs have a wide range of medicinal properties, including antibacterial and anticancer effects. Here, the authors explored a library of alkyl or aryl berberine analogs to probe binding to double-stranded and G-quadruplex DNA. They determined that the nature of the substituent, the position of the substituent, and the nucleic acid target affect the free energy of binding of berberine analogs to DNA and G-quadruplex DNA, however berberine analogs did not result in net stabilization of G-quadruplex DNA.
Read More...Estimating the liquid jet breakdown height using dimensional analysis with experimental evidence
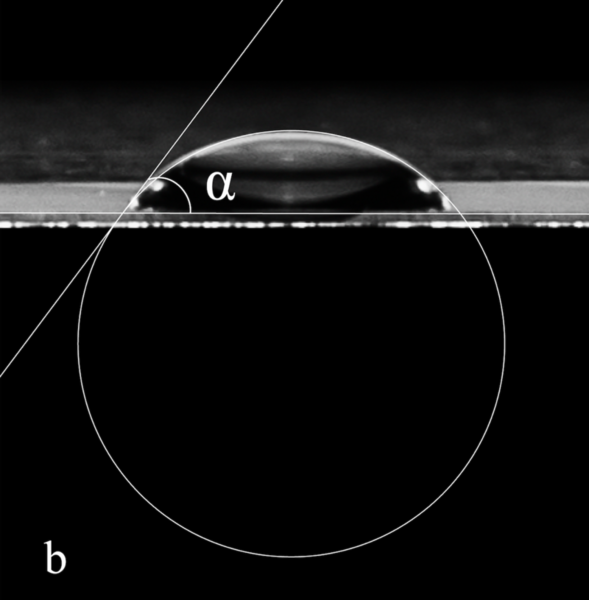
These authors mathematically deduce a model that explains the interesting (and unintuitive) physical phenomenon that occurs when water falls.
Read More...Quantitative NMR spectroscopy reveals solvent effects in the photochemical degradation of thymoquinone
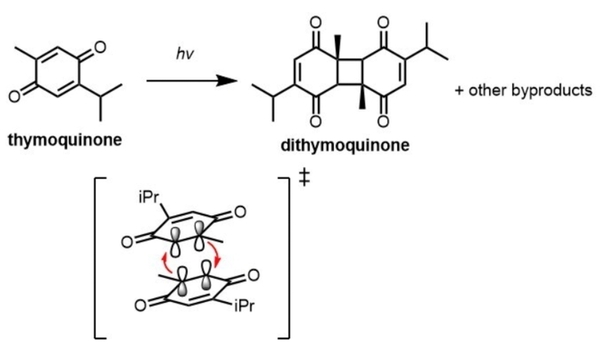
Thymoquinone is a compound of great therapeutic potential and scientific interest. However, its clinical administration and synthetic modifications are greatly limited by its instability in the presence of light. This study employed quantitative 1H nuclear magnetic resonance (NMR) spectroscopy to identify the effect of solvation on the degradation of thymoquinone under ultraviolet light (UV). It found that the rate of degradation is highly solvent dependent occurs maximally in chloroform.
Read More...Comparative singlet oxygen photosensitizer efficiency of berberine, rose bengal, and methylene blue by time course nuclear magnetic resonance (NMR) monitoring of a photochemical 4+2 cycloaddition endoperoxide formation

Berberine, a natural product alkaloid, has been shown to exert biological activity via in situ production of singlet oxygen when photo irradiated. Berberine utilizes singlet oxygen in its putative mechanism of action, wherein it forms an activated complex with DNA and photosensitizes triplet oxygen to singlet oxygen to specifically oxidize guanine residues, thereby halting cell replication and leading to cell death. This has potential application in photodynamic therapy, alongside other such compounds which also act as photosensitizers and produce singlet oxygen in situ. The quantification of singlet oxygen in various photosensitizers, including berberine, is essential for determining their photosensitizer efficiencies. We postulated that the singlet oxygen produced by photoirradiation of berberine would be superior in terms of singlet oxygen production to the aforementioned photosensitizers when irradiated with UV light, but inferior under visible light conditions, due to its strong absorbance of UV wavelengths.
Read More...High-performance liquid chromatography insight in pH-dependent hydrolysis of andrographolide acetonide
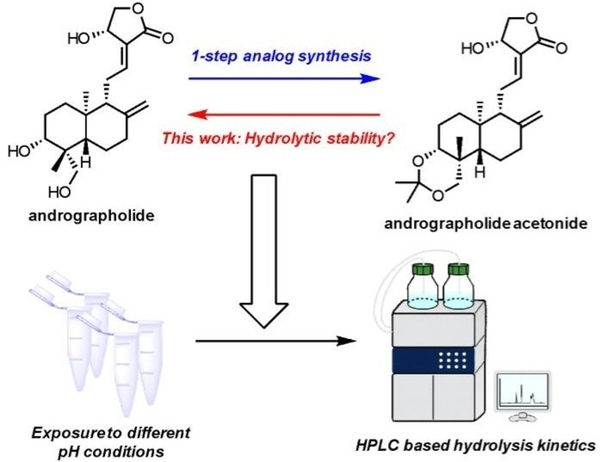
Andrographolide, a natural compound with anti-inflammatory, antidepressant, and anti-cancer properties, can be chemically modified by adding an acetonide group to form andrographolide acetonide, which is more potent and acts as a pH-dependent prodrug. Researchers investigated the hydrolysis of this acetonide group under mildly acidic conditions.
Read More...Deuterated solvent effects in the kinetics and thermodynamics of keto-enol tautomerization of ETFAA
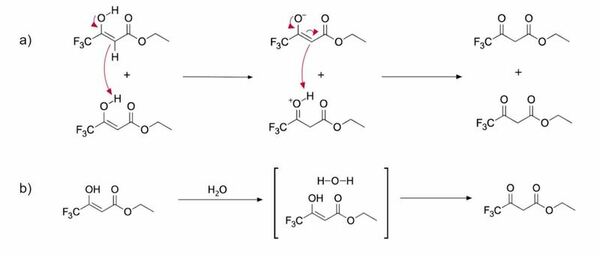
In this study, the authors determined whether tautomerization dynamics in protic and aprotic solvents displayed differences in reaction rates and in the proportion of the keto and enol tautomers present.
Read More...Strain selective in vitro and in silico structure activity relationship of tetracycline antibiotics
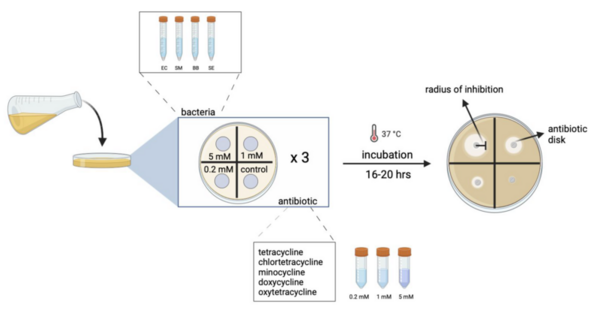
The authors looked at four members of the tetracycline antibiotic family/class against four different species of bacteria.
Read More...Spider Density Shows Weak Relationship with Vegetation Density

Evidence supports that spiders have many ecological benefits including insect control and predation in the food chain. In this study the authors investigate that whether the percent of vegetation coverage and spider density are correlated. They determine that despite the trend there is no statistically significant correlation.
Read More...Exploration of the density–size correlation of celestial objects on various scales
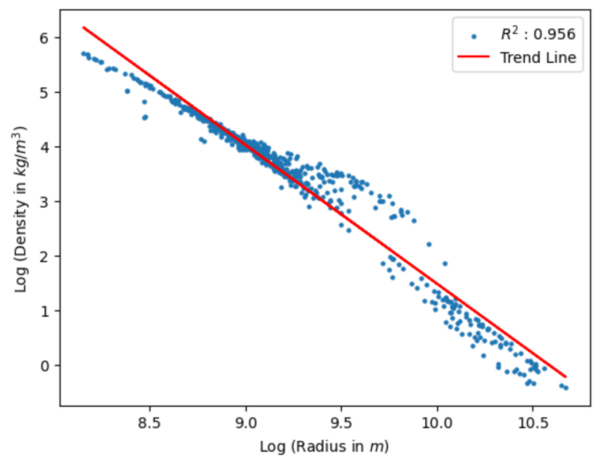
Building on previous work by earlier astronomers, the authors investigate the correlation between the density and size of celestial objects in the universe, including neutron stars, galaxies, and galaxy clusters.
Read More...