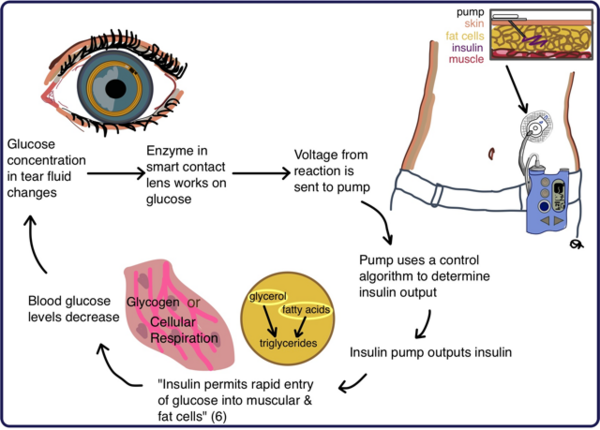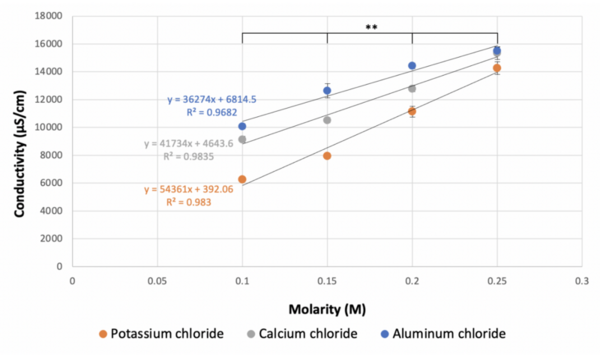
The authors looked at how molarity impacts the degree to which ionic compounds dissociate in solution. They found that lower molarities led to decreased conductivity of solutions in a manner that did not follow the theoretical predictions.
Read More...