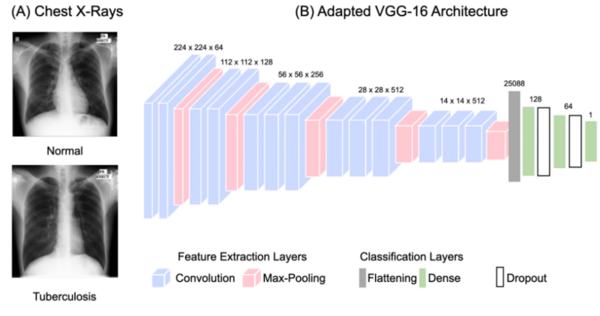
The authors developed and tested machine learning methods to diagnose tuberculosis from pulmonary X-ray scans.
Read More...Effects of data amount and variation in deep learning-based tuberculosis diagnosis in chest X-ray scans

The authors developed and tested machine learning methods to diagnose tuberculosis from pulmonary X-ray scans.
Read More...The effects of image manipulation on classification of cervical spondylosis X-ray images using deep learning
SmartZoo: A Deep Learning Framework for an IoT Platform in Animal Care
Zoos offer educational and scientific advantages but face high maintenance costs and challenges in animal care due to diverse species' habits. Challenges include tracking animals, detecting illnesses, and creating suitable habitats. We developed a deep learning framework called SmartZoo to address these issues and enable efficient animal monitoring, condition alerts, and data aggregation. We discovered that the data generated by our model is closer to real data than random data, and we were able to demonstrate that the model excels at generating data that resembles real-world data.
Read More...A novel deep learning model for visibility correction of environmental factors in autonomous vehicles

Intelligent vehicles utilize a combination of video-enabled object detection and radar data to traverse safely through surrounding environments. However, since the most momentary missteps in these systems can cause devastating collisions, the margin of error in the software for these systems is small. In this paper, we hypothesized that a novel object detection system that improves detection accuracy and speed of detection during adverse weather conditions would outperform industry alternatives in an average comparison.
Read More...Impact of length of audio on music classification with deep learning
The authors looked at how the length of an audio clip used of a song impacted the ability to properly classify it by musical genre.
Read More...Forecasting air quality index: A statistical machine learning and deep learning approach

Here the authors investigated air quality forecasting in India, comparing traditional time series models like SARIMA with deep learning models like LSTM. The research found that SARIMA models, which capture seasonal variations, outperform LSTM models in predicting Air Quality Index (AQI) levels across multiple Indian cities, supporting the hypothesis that simpler models can be more effective for this specific task.
Read More...The Role of a Mask - Understanding the Performance of Deep Neural Networks to Detect, Segment, and Extract Cellular Nuclei from Microscopy Images
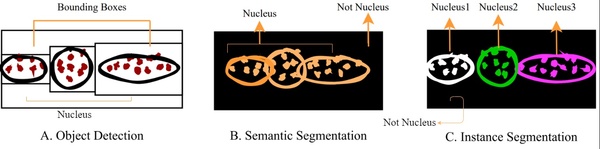
Cell segmentation is the task of identifying cell nuclei instances in fluorescence microscopy images. The goal of this paper is to benchmark the performance of representative deep learning techniques for cell nuclei segmentation using standard datasets and common evaluation criteria. This research establishes an important baseline for cell nuclei segmentation, enabling researchers to continually refine and deploy neural models for real-world clinical applications.
Read More...A comparative analysis of machine learning approaches for prediction of breast cancer
Machine learning and deep learning techniques can be used to predict the early onset of breast cancer. The main objective of this analysis was to determine whether machine learning algorithms can be used to predict the onset of breast cancer with more than 90% accuracy. Based on research with supervised machine learning algorithms, Gaussian Naïve Bayes, K Nearest Algorithm, Random Forest, and Logistic Regression were considered because they offer a wide variety of classification methods and also provide high accuracy and performance. We hypothesized that all these algorithms would provide accurate results, and Random Forest and Logistic Regression would provide better accuracy and performance than Naïve Bayes and K Nearest Neighbor.
Read More...Collaboration beats heterogeneity: Improving federated learning-based waste classification

Based on the success of deep learning, recent works have attempted to develop a waste classification model using deep neural networks. This work presents federated learning (FL) for a solution, as it allows participants to aid in training the model using their own data. Results showed that with less clients, having a higher participation ratio resulted in less accuracy degradation by the data heterogeneity.
Read More...Transfer learning and data augmentation in osteosarcoma cancer detection
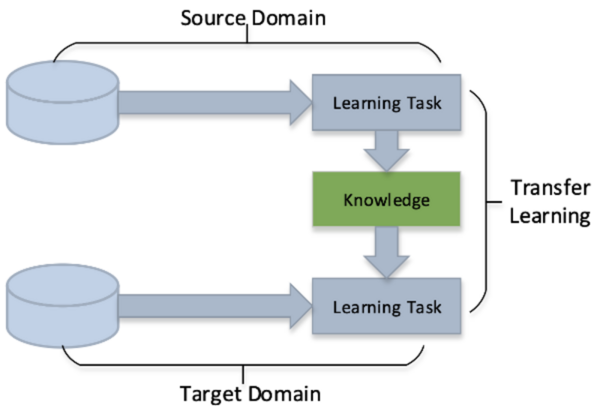
Osteosarcoma is a type of bone cancer that affects young adults and children. Early diagnosis of osteosarcoma is crucial to successful treatment. The current methods of diagnosis, which include imaging tests and biopsy, are time consuming and prone to human error. Hence, we used deep learning to extract patterns and detect osteosarcoma from histological images. We hypothesized that the combination of two different technologies (transfer learning and data augmentation) would improve the efficacy of osteosarcoma detection in histological images. The dataset used for the study consisted of histological images for osteosarcoma and was quite imbalanced as it contained very few images with tumors. Since transfer learning uses existing knowledge for the purpose of classification and detection, we hypothesized it would be proficient on such an imbalanced dataset. To further improve our learning, we used data augmentation to include variations in the dataset. We further evaluated the efficacy of different convolutional neural network models on this task. We obtained an accuracy of 91.18% using the transfer learning model MobileNetV2 as the base model with various geometric transformations, outperforming the state-of-the-art convolutional neural network based approach.
Read More...