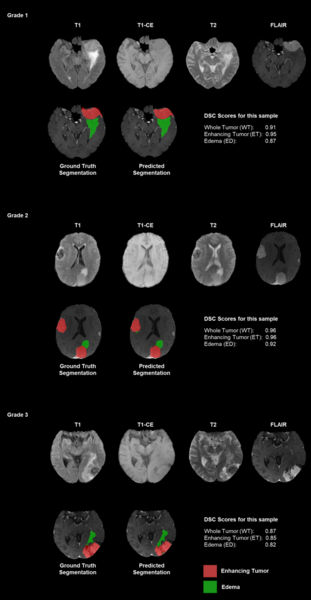
Authors emphasize the challenges of manual tumor segmentation and the potential of deep learning models to enhance accuracy by automatically analyzing MRI scans.
Read More...Evaluating the clinical applicability of neural networks for meningioma tumor segmentation on 3D MRI

Authors emphasize the challenges of manual tumor segmentation and the potential of deep learning models to enhance accuracy by automatically analyzing MRI scans.
Read More...The design of Benzimidazole derivatives to bind to GDP-bound K-RAS for targeted cancer therapy
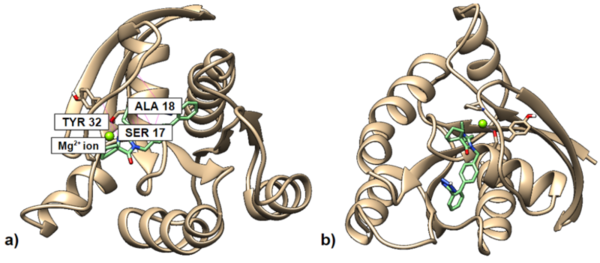
In this study, the authors looked at a proto-oncogene, KRAS, and searched for molecules that are predicted to be able to bind to the inactive form of KRAS. They found that a modified version of Irbesartan, a derivative of benzimidazole, showed the best binding to inactive KRAS.
Read More...DyGS: A Dynamic Gene Searching Algorithm for Cancer Detection
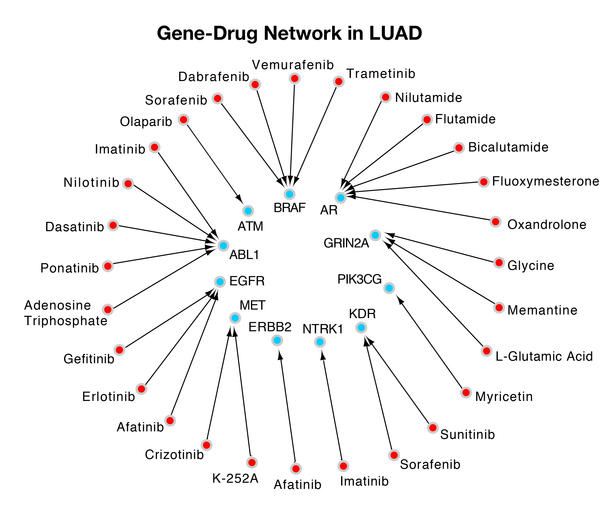
Wang and Gong developed a novel dynamic gene-searching algorithm called Dynamic Gene Search (DyGS) to create a gene panel for each of the 12 cancers with the highest annual incidence and death rate. The 12 gene panels the DyGS algorithm selected used only 3.5% of the original gene mutation pool, while covering every patient sample. About 40% of each gene panel is druggable, which indicates that the DyGS-generated gene panels can be used for early cancer detection as well as therapeutic targets in treatment methods.
Read More...Applying machine learning to breast cancer diagnosis: A high school student’s exploration using R
The authors combine fine needle aspiration biopsy and machine learning algorithms to develop a breast cancer detection method suitable for resource-constrained regions that lack access to mammograms.
Read More...Citrate and lactate drive glioblastoma progression via activation of tumor-associated macrophages
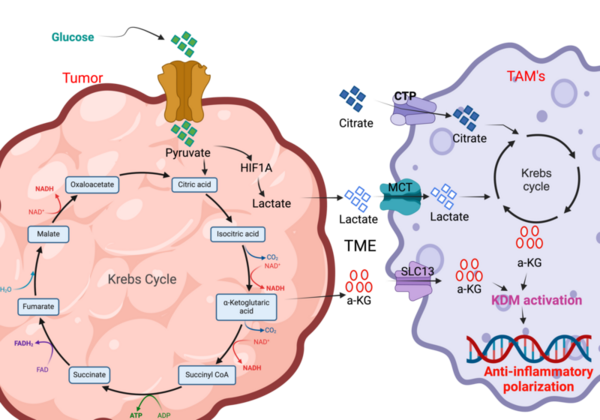
The authors looked at the impact of citrate and lactate on glioblastoma progression. Their results provide important insights for future immunotherapies aimed at treating glioblastoma.
Read More...How artificial intelligence deep learning models can be used to accurately determine lung cancers
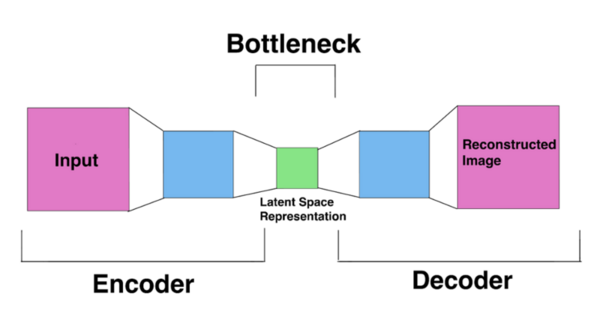
The authors looked at the ability of different deep learning models to predict the presence of lung cancer from chest CT scans. They found that a pre-trained CNN model performed better than an autoencoder model.
Read More...Advancements in glioma segmentation: comparing the U-Net and DeconvNet models
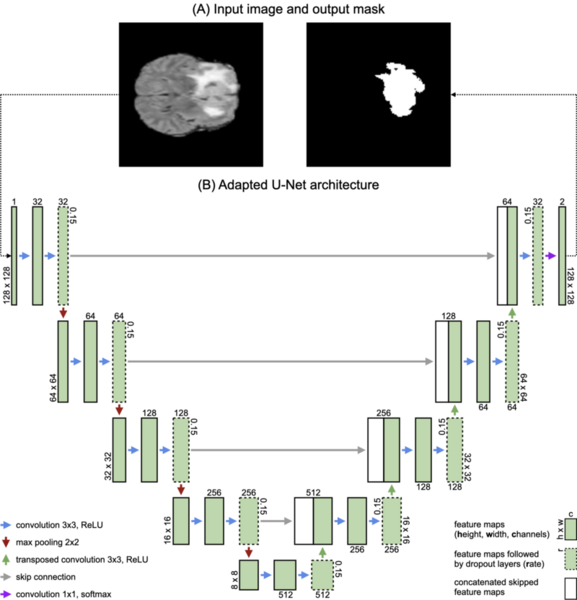
This study compares the performance of two deep learning models, U-Net and DeconvNet, for segmenting gliomas from MRI scans.
Read More...Discovery of novel targets for diffuse large B-cell lymphoma
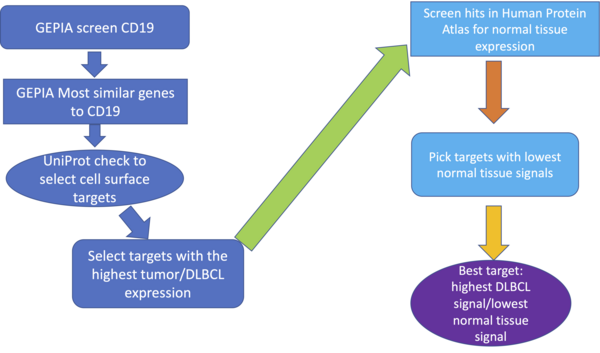
In this study, the authors identify new potential targets to treat advanced diffuse large B-cell lymphoma after treatment relapse and loss of CD19 expression.
Read More...Significance of Tumor Growth Modeling in the Behavior of Homogeneous Cancer Cell Populations: Are Tumor Growth Models Applicable to Both Heterogeneous and Homogeneous Populations?
This study follows the process of single-cloning and the growth of a homogeneous cell population in a superficial environment over the course of six weeks with the end goal of showing which of five tumor growth models commonly used to predict heterogeneous cancer cell population growth (Exponential, Logistic, Gompertz, Linear, and Bertalanffy) would also best exemplify that of homogeneous cell populations.
Read More...A Novel Approach to Prevent and Restrict Early Stages of Cancer Cell Growth Using a Combination of Moringa and Sesame in a Drosophila Model

Sesame (Sesamum indicum) and moringa (Moringa oleifera) have natural antioxidants that could prevent cancer growth. Previously, this group found that sesame and moringa individually suppress eye tumor grown in the Drosophila melanogaster model. In the present study, combinations of sesame and moringa at different concentrations were included in the D. melanogaster diet. The impact on eye tumor development was assessed at different stages of growth.
Read More...