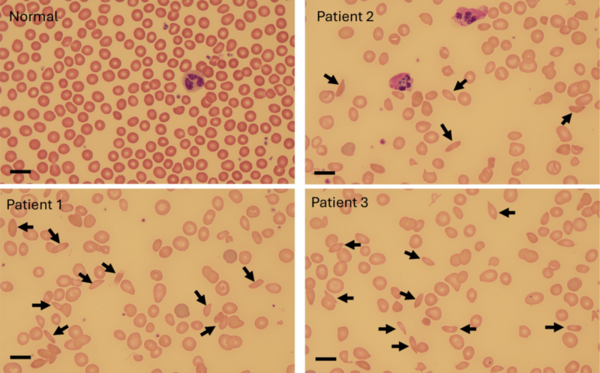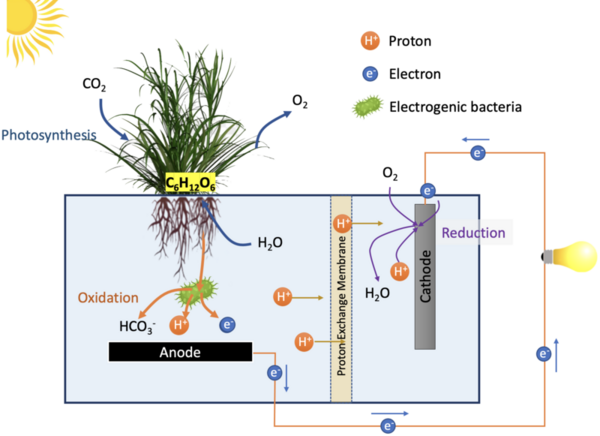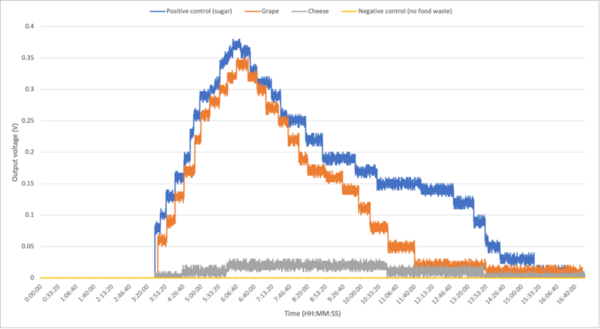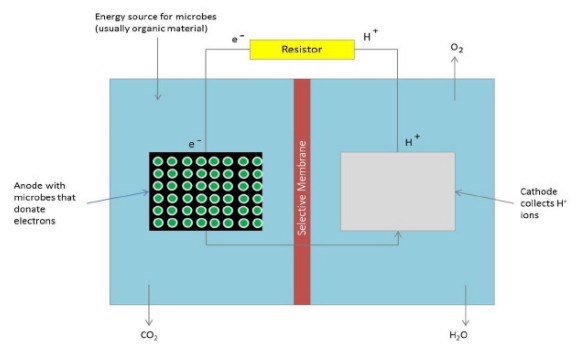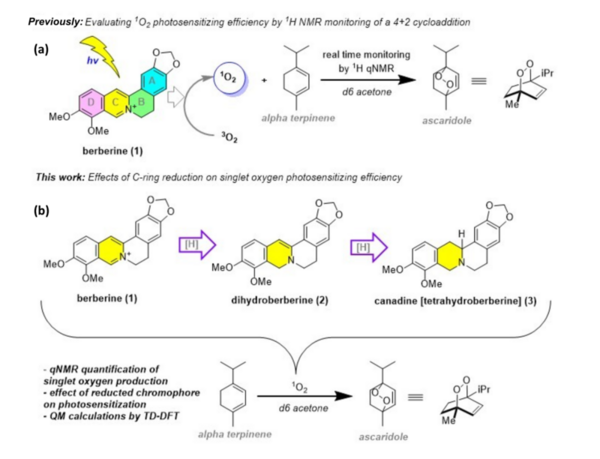
Berberine is a natural product isoquinoline alkaloid derived from plants of the genus Berberis. When exposed to photoirradiation, it produces singlet oxygen through photosensitization of triplet oxygen. Through qNMR analysis of 1H NMR spectra gathered through kinetic experiments, we were able to track the generation of a product between singlet oxygen and alpha terpinene, allowing us to quantitatively measure the photosensitizing properties of our scaffolds.
Read More...