
The authors tested the impact of various additives on biopolymer degradation.
Read More...A comparison of starches and plasticizers for biopolymer synthesis and degradation
The effect of molecular weights of chitosan on the synthesis and antifungal effect of copper chitosan
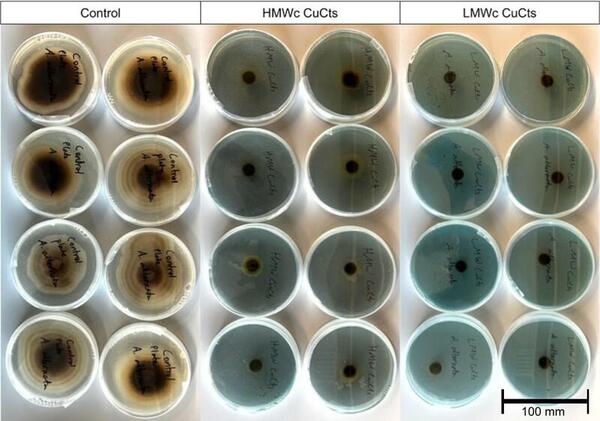
Pathogenic fungi such as Alternaria alternata (A. alternata) can decimate crop yields and severely limit food supplies when left untreated. Copper chitosan (CuCts) is a promising alternative fungicide for developing agricultural areas due to being inexpensive and nontoxic. We hypothesized that LMWc CuCts would exhibit greater fungal inhibition due to the beneficial properties of LMWc.
Read More...Which fruit peel helps retain the most soil moisture?

Here, the authors investigated the ability to use fruit peels to help soil retain moisture, a property that is essential to agriculture. Across a 96-hour observation period, orange, banana, and kiwi peel water emulsions were evaluated for their effects on soil moisture. They found that orange peels retained the most moisture, but banana and kiwi peels also offered improvements over their control sample.
Read More...Developing “Off the Shelf” Pancreases for Diabetic Patients Using Bacterial and Kombucha Tea Waste
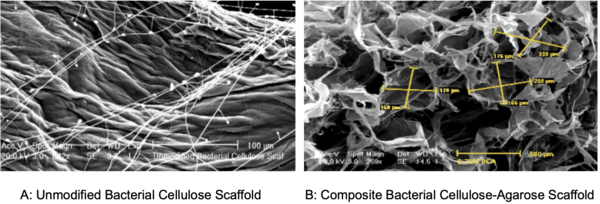
In this study, the authors investigate the suitability of using bacterial cellulose as a scaffold for cell transplants. Interestingly, this cellulose is a can be found in the discard from a symbiotic culture of bacteria and yeast (SCOBY) used to make kombucha.
Read More...A Taste of Sweetness in Bioplastics

Sweet potatoes are one of the most common starches in Taiwan, and sweet potato peels hold significant potential to make biodegradable plastics which can alleviate the environmental impact of conventional petroleum-based plastics. In this paper, Tsai et al created starch-based bioplastics derived from sweet potato peels and manipulated the amount of added glycerol to alter the plastic’s strength and flexibility properties. Their results indicated that higher concentrations of glycerol yield more malleable plastics, providing insights into how recycled agricultural waste material might be used to slow down the rate of pollution caused by widespread production of conventional plastics.
Read More...Understanding the Mechanism of Star-Block Copolymers as Nanoreactors for Synthesis of Well-Defined Silver Nanoparticles
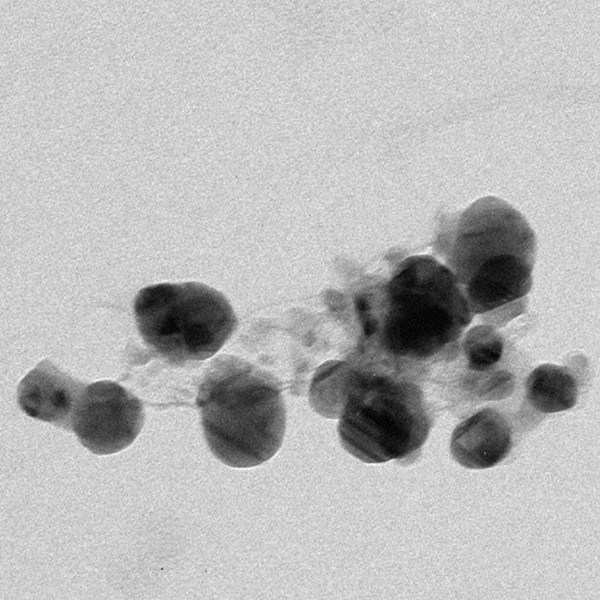
Here, the authors characterize how silver ions nucleate a star-block copolymer to generate nano-sized silver particles.
Read More...Covalently Entrapping Catalase into Calcium Alginate Worm Pieces Using EDC Carbodiimide as a Crosslinker.
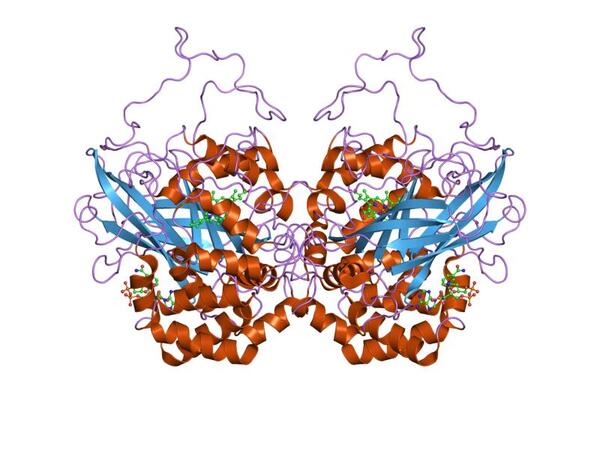
Catalase is a biocatalyst used to break down toxic hydrogen peroxide into water and oxygen in industries such as cheese and textiles. Improving the efficiency of catalase would help us to make some industrial products, such as cheese, less expensively. The best way to maintain catalase’s conformation, and thus enhance its activity, is to immobilize it. The primary goal of this study was to find a new way of immobilizing catalase.
Read More...Sepia bandensis ink inhibits polymerase chain reactions
_(35622760083).jpg)
While cephalopods play significant roles in both ecosystems and medical research, there is currently no assembled genome. In an attempt to sequence the Sepia bandensis genome, it was found that there was inhibition from the organism during DNA extraction, resulting in PCR failure. In this study, researchers tested the hypothesis that S. bandensis ink inhibits PCR. They then assessed the impact of ink on multiple methods of DNA extraction
Read More...The Development of a Superhydrophobic Surface Using Electrolytic Deposition & Polymer Chains Precipitation
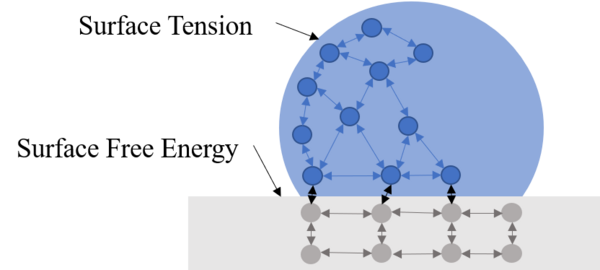
In this study, the authors were interested in developing a hydrophobic surface that will extend the lifespan of metals by reducing water exposure and other damage. The used a zinc coating on steel to pursue this effort.
Read More...The Effects of Post-Consumer Waste Polystyrene on the Rate of Mealworm Consumption

In a world where plastic waste accumulation is threatening both land and sea life, Green et al. investigate the ability of mealworms to breakdown polystyrene, a non-recyclable form of petrochemical-based polymer we use in our daily lives. They confirm that these organisms, can degrade various forms of polystyrene, even after it has been put to use in our daily lives. Although the efficiency of the degradation process still requires improvement, the good news is, the worms are tiny and themselves are biodegradable, so we can use plenty of them without worrying about space and how to get rid of them. This is very promising and certainly good news for the planet.
Read More...