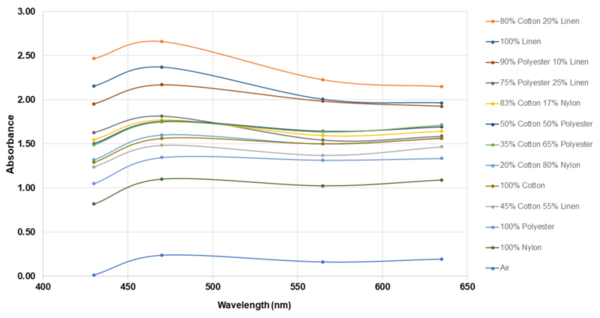
The authors looked at how different blends of synthetic and non-synthetic fabrics protected against UV radiation.
Read More...Effectiveness of different fabrics in protecting from ultraviolet rays

The authors looked at how different blends of synthetic and non-synthetic fabrics protected against UV radiation.
Read More...Improper storage of sunscreen might decrease effectiveness

This study explored sunscreen storage temperature affects the efficacy of sunscreen to block UV light.
Read More...The decision-making process of an MLB batter: effects of feedback stimuli and anxiety on batting performance

This study examined whether findings from laboratory studies of baseball cognition also apply during real games. Using 2024 MLB data from 30 players, researchers analyzed batting performance under different levels of feedback and anxiety.
Read More...Algorithmic barriers: Investigating student perceptions of AI bias in subjective “culture fit” hiring

This study investigated perceptions of the emerging workforce toward the use of artificial intelligence in hiring, specifically for assessing subjective "culture fit." Through a mixed-methods survey of 150 high school and early-college students in Nepal, we found a significant disconnect between organizational adoption of AI and the profound skepticism of young job candidates, who express deep concerns about fairness, transparency, and the potential for AI to perpetuate systemic discrimination.
Read More...Leveraging transfer learning with convolutional neural networks for cardiovascular disease detection
This study shows the efficacy of leveraging transfer learning, specifically from residual networks, to detect CVDs and possible signs of CVDs. The findings indicate that leveraging transfer learning from residual networks alongside medical professionals is a highly promising approach for CVD detection and diagnosis, warranting further investigation.
Read More...The effects of a high-sucrose diet on the survival of Drosophila melanogaster from a bacterial infection
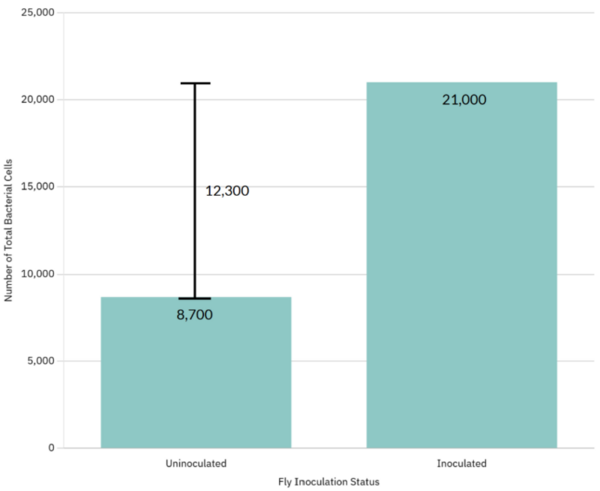
Excess sucrose consumption has been associated with several health problems, including inflammation and potential negative effects on immune function. However, the exact relationship between sucrose intake and immunity remains unclear, especially during bacterial infections. This study examined how sucrose intake affected the survival of fruit flies following oral infection with the bacterial pathogen Serratia marcescens.
Read More...The effect of chondroitin sulfate on the development of plants grown in roadside soil
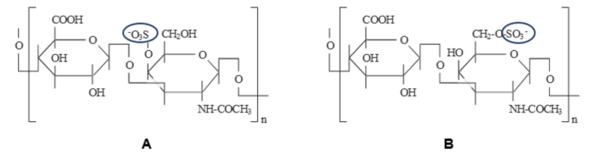
Heavy metal contamination from transportation can damage soil and vegetation by increasing oxidative stress, disrupting enzymes, and impairing photosynthesis. This study investigated whether chondroitin sulfate (CS) could reduce the harmful effects of contaminated roadside soil on plant growth by binding with metal ions.
Read More...Eye color, visual acuity and photophobia: How eye color affects light sensitivity
This study examined whether eye color affects photophobia and vision in elementary school students and staff, finding no significant relationship between eye color, light sensitivity, or visual acuity. However, photophobia was common across age groups, highlighting the need for greater awareness of light sensitivity in learning environments.
Read More...Too hot to work? Heat waves, household income, and labor adaptation in India

Paper found that heat waves in India are linked to lower household income, agricultural income, and consumption, with agriculture being affected the most. It also suggests farm workers may adapt to extreme heat over time by increasing labor inputs despite rising temperatures.
Read More...Exploring Interactions between PFAS (Per- and Polyfluoroalkyl Substances) and proteins
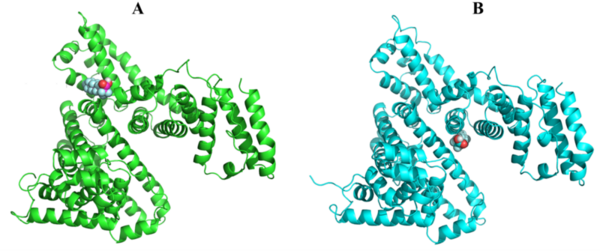
Here the authors investigated how the "forever chemical" perfluorooctanoic acid binds to bovine serum albumin (BSA) using computational software to simulate its potential impact on essential human plasma proteins. They identify a possible, high-energy binding configuration that could persistently impair protein functions, underscoring the critical need for further research into the long-term health risks of per- and poly-fluoroalkyl substances exposure.
Read More...