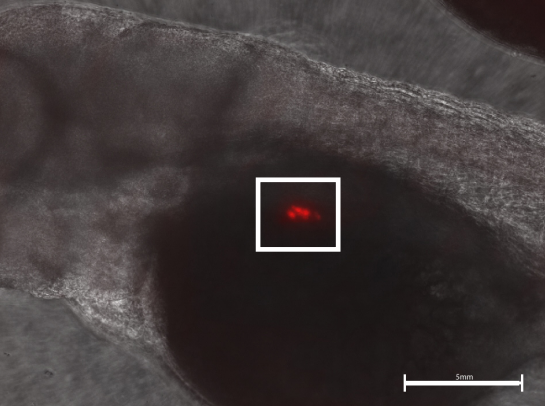
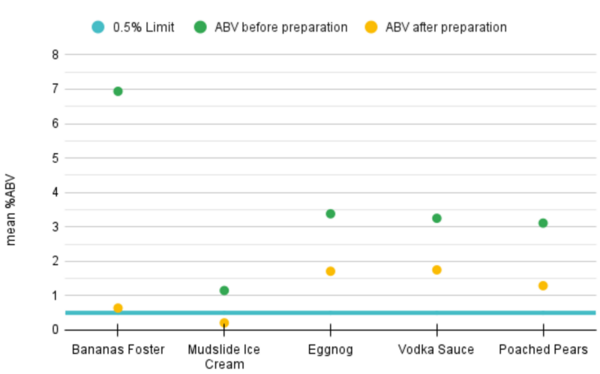
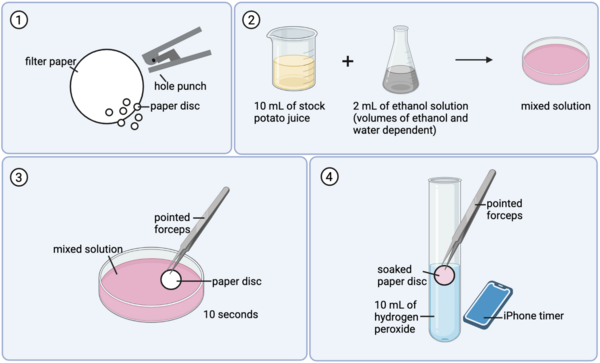
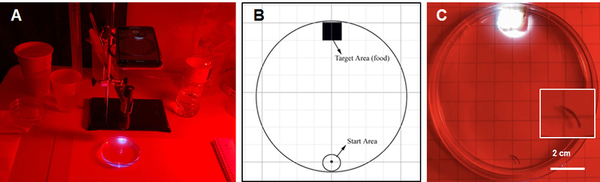
Alcohol use disorder is a chronic, relapsing disease that affects millions of Americans every day. There are limited treatment options for alcohol dependence and alcohol withdrawal symptoms, including depression and anxiety. Previous studies have shown that probiotics can decrease depression in rodents during maternal separation and anxiety in humans. Therefore, we hypothesized that the ethanol-withdrawn planaria who consumed probiotics would have decreased withdrawal symptoms as measured by increased motility compared to the ethanol-withdrawn planaria that were not fed probiotics. The ethanol-withdrawn planaria had a statistically significant decrease in motility compared to the control group, while the planaria that consumed probiotics had no statistically significant change in motility compared to the control group.
Read More...