Exploring Interactions between PFAS (Per- and Polyfluoroalkyl Substances) and proteins
(1) BASIS Independent McLean, (2) Nanjing University
https://doi.org/10.59720/25-038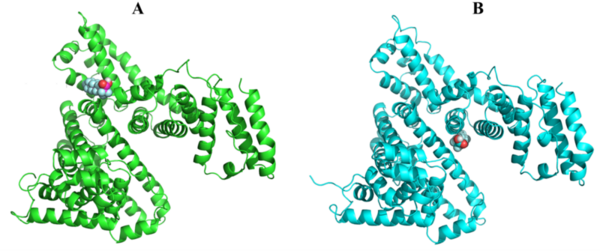
PFAS, or per- and poly-fluoroalkyl substances, while not as infamous or widely discussed as microplastics, are among the biggest threats to human health and development. Like microplastics, PFAS take an extremely long time to degrade, earning them the title of the “forever chemicals.” We investigated the potential dangers of PFAS by focusing on how perfluorooctanoic acid (PFOA), one of the most studied PFAS, binds to bovine serum albumin (BSA), and how this interaction could impact protein function and human health. We used BSA because of its structural similarity to human serum albumin (HSA), the most abundant protein in adult plasma, making it integral to bodily functions. We hypothesized that PFOA would bind with high affinity to BSA, causing structural changes that might affect the protein's functions. Using several software programs, we examined two distinct configurations of the PFOA-BSA complex. We identified two configurations with differing properties, one having a greater stability than the other. However, despite having less stability, the second configuration had a higher binding energy, implying a stronger and more likely binding scenario in the human body. This may contribute to long-term health issues due to persistent PFAS binding, potentially impairing protein functions and leading to various health problems. Our study highlights the need for further research to understand these interactions, which could lead to strategies to address the health risks posed by PFAS exposure.
This article has been tagged with: