The Cilium- and Centrosome-Associated Protein CCDC11 Is Required for Cytokinesis via Midbody Recruitment of the ESCRT- III Membrane Scission Complex Subunit CHMP2A
(1) Half Hollow Hills High School, Dix Hills, New York, (2) Department of Pharmacological Sciences, Stony Brook University, Stony Brook, New York, (3) Department of Biochemistry and Cell Biology, Stony Brook University, Stony Brook, New York
https://doi.org/10.59720/17-013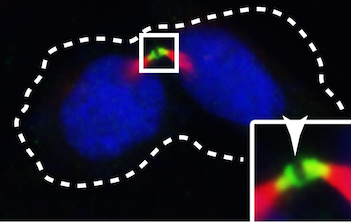
Cell division is a fundamental biological process, crucial for the normal development of an organism as well as tissue repair, regeneration, and wound healing. During cell division, there are multiple critical checkpoints that ensure accurate replication and distribution of DNA. The final stage, cytokinesis, involves the physical separation of two newly formed daughter cells. Our research focused on elucidating the molecular functions of the coiled-coil domain-containing 11 (CCDC11) protein, which has been associated with human laterality disorders and plays critical roles in the formation and function of cilia, hair- like structures on the apical cell surface. We found that, in addition to cilia, CCDC11 localizes to the cleavage furrow and midbody between two dividing daughter cells of various human cell lines. CCDC11 contains three coiled-coil (CC) domains, and the CC1 and CC2 domains are responsible for its midbody localization. We also observed that depletion of CCDC11 results in a significant increase in the number of multinucleated cells, indicating defects in cytokinesis, the final stage of cell division. Furthermore, CCDC11-depleted cells exhibit diminished midbody recruitment of the protein CHMP2A, a critical component of the ESCRT-III membrane scission machinery. Collectively, our findings uncover a novel and previously unreported role for CCDC11 in cytokinesis.
This article has been tagged with: