Ribosome distribution affects stalling in amino-acid starved cancer cells
(1) Las Lomas High School, Walnut Creek, California, (2) Princeton University, Princeton, New Jersey
https://doi.org/10.59720/20-215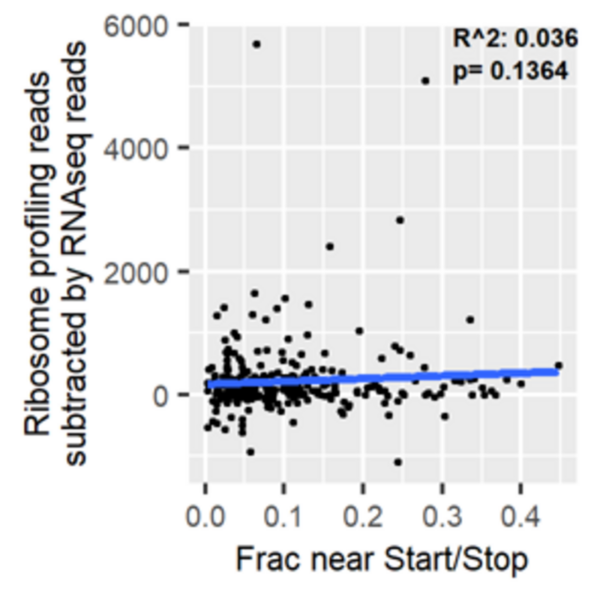
Protein synthesis is a process central to all life on Earth, including mammalian cells. During this process, ribosomes attach to mRNA strands and translate them into proteins using amino acids. Under stress (for example, when the supply of circulating amino acids has been disrupted by tissue injury), ribosomes can stall. Ribosome profiling, a technique that creates a snapshot of active ribosomes in a cell by sequencing ribosome-protected mRNA fragments, captures a snapshot of ribosomes along transcripts and can detect such stalling events. This method has also revealed that the patterns of ribosomes along transcripts can vary from transcript to transcript in a manner that has not yet been explained. Here, we analyzed ribosome profiling data from amino acid-starved pancreatic cancer cells to explore whether the pattern of ribosome distribution along transcripts under normal conditions can predict the degree of ribosome stalling under stress. We hypothesized that ribosomes would stall more along “elongation-limited” transcripts that have fewer ribosome footprints near the start and stop codons than “initiation-limited” transcripts that have a large fraction of footprints at the start codon. Indeed, we found that ribosomes in amino acid-deprived cells stalled more along elongation-limited transcripts. By contrast, we observed no relationship between read density near start and stop and disparities between mRNA sequencing reads and ribosome profiling reads. This research identifies an important relationship between read distribution and propensity for ribosomes to stall, although more work is needed to fully understand the patterns of ribosome distribution along transcripts in ribosome profiling data.
This article has been tagged with: