Investigating the Role of the Novel ESCRT-III Recruitment Factor CCDC11 in HIV Budding: A Potential Target for Antiviral Therapy
(1) Ward Melville High School, East Setauket, New York, (2) Half Hollow Hills High School West, Dix Hills, New York, (3) Department of Biochemistry and Cell Biology, Stony Brook University, Stony Brook, New York, (4) Department of Microbiology and Immunology, Stony Brook University, Stony Brook, New York, (5) Department of Pharmacological Sciences, Stony Brook University, Stony Brook, New York
https://doi.org/10.59720/19-109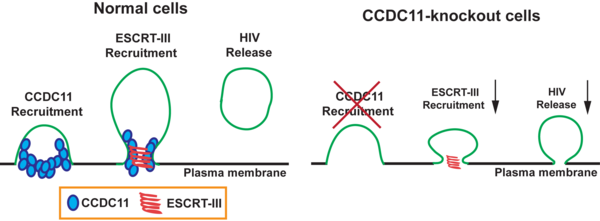
Acquired immunodeficiency syndrome (AIDS) is a life-threatening condition caused by the human immunodeficiency virus (HIV). By damaging the immune system, HIV compromises the body's ability to fight infectious and non-infectious diseases. To discover potential targets for antiviral therapies, we explored the role of Coiled-Coil Domain-Containing 11 (CCDC11) in HIV-1 budding. CCDC11 serves an important role in cytokinesis by recruiting Endosomal Sorting Complex Required for Transport III (ESCRT-III) membrane scission machinery to the midbody of the cell in preparation for the separation into two daughter cells. ESCRT-III has also been implicated in the budding process of HIV and other enveloped viruses. Therefore, we hypothesized that CCDC11 is required for viral budding. To investigate this, we performed ELISAs (enzyme-linked immunosorbent assays) to detect the HIV-1 Gag capsid protein p24 as a measure of viral particle release. We found that ectopic expression of CCDC11 in human embryonic kidney (HEK) 293T cells significantly increased the production of HIV-1 particles in culture media. To rigorously test our hypothesis, we established CCDC11-knockout HEK293T cells using CRISPR-Cas9 technology. CCDC11 knockout was verified by DNA sequencing, western blotting, and immunofluorescence staining. We found that knockout of CCDC11 markedly decreased the production of HIV-1 particles. The defective viral production in CCDC11-knockout cells was partially restored when wild-type CCDC11 was re-expressed. Collectively, our data suggested that CCDC11 is critical for efficient HIV-1 budding. Considering that CCDC11 expression is generally low in the majority of adult human tissues, CCDC11 might be a viable target for antiviral therapeutics without major side effects.
This article has been tagged with: