The Effects of L-glutamate, L-glutamine, and L-aspartic Acid on the Amylase Production of E. coli Transformed With pAmylase
(1) Wilde Lake High School, Howard County, Maryland
https://doi.org/10.59720/15-081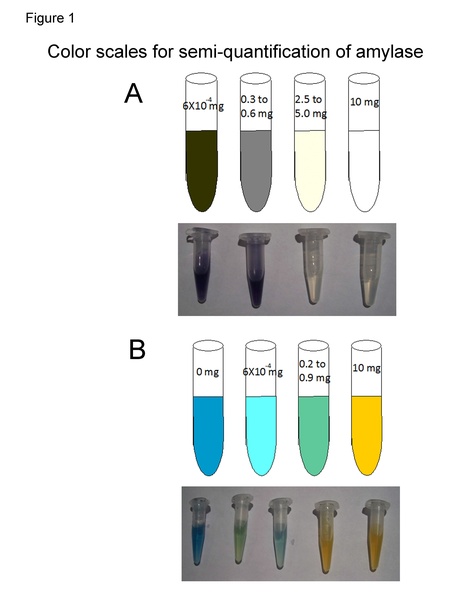
Amylase catalyzes the hydrolysis of dietary starch into oligosaccharides. In humans, amylase is produced by the pancreas and salivary glands and is important in food digestion. Human amylase also has broad applications for therapeutic use in patients with pancreatic insufficiency, necessitating an efficient process for amylase production. E. coli transfected with human amylase cDNA can be used to produce amylase to fulfill these pharmaceutical production needs. It was reported that the production of Thermus maltogenic amylase in E. coli is enhanced by adding L-glutamate and L-glutamine. The purpose of this experiment was to determine whether the amino acids L-glutamate, L-glutamine, and L-aspartic acid could increase human amylase expression in E. coli. Based on their effect on the production of Thermus maltogenic amylase in E. coli, we hypothesized that L-glutamate and L-glutamine would increase human amylase production in E. coli, while L-aspartic acid would not. Bacteria with added L-glutamate produced 16-fold more amylase, according to Lugol’s iodine staining, and 52-fold more amylase according to Benedict’s solution. Bacteria with L-glutamine produced 6-fold more amylase, according to Lugol’s iodine staining, and 26-fold more amylase according to Benedict’s solution. These findings demonstrate L-glutamate and L-glutamine’s ability to increase amylase production by increasing the bacterial cell population. This may result from the effect of L-glutamate and L-glutamine’s nutritional properties, as well as their roles as precursors to amino acid synthesis. Our study presents preliminary results for the further development of a more productive way to produce amylase for the pharmaceutical industries.
This article has been tagged with: