De novo design of a dual-target inhibitor against tau phosphorylation and acetylation for Alzheimer's therapy
(1) Plano West Senior High School
https://doi.org/10.59720/25-250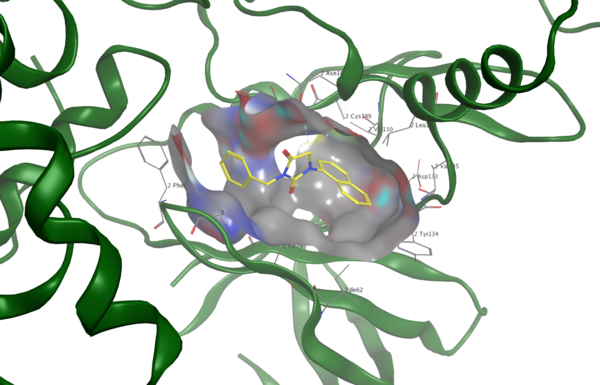
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder affecting approximately 7 million Americans and is the most common form of dementia. The pathological hallmarks of AD are the accumulation of amyloid-beta plaques and neurofibrillary tangles composed of tau protein that have undergone abnormal post-translational modifications (PTMs). While tau hyperphosphorylation has been extensively studied, tau acetylation occurs at fewer sites and remains comparatively understudied, representing a critical gap in understanding AD progression. In this study, we investigated how tau acetylation influences aggregation relative to phosphorylation and evaluated whether a brain-permeable retro-inverso peptide could be rationally designed to inhibit both glycogen synthase kinase-3β (GSK3β), a major tau kinase, and the tau-acetylating enzyme p300. We hypothesized that acetylation would promote aggregation less strongly than phosphorylation due to its lower reported prevalence. To test this, we computationally modeled full-length human tau and generated native, phosphorylated, acetylated, and dual-modified variants at AD-associated residues. Aggregation propensity and structural flexibility analyses revealed that acetylation significantly increased aggregation and conformational instability, comparable to phosphorylation, while dual modification produced additive destabilizing effects. We then designed a retro-inverso peptide inhibitor, RI-GSK, and evaluated its binding to GSK3β and p300 using molecular docking. RI-GSK demonstrated stronger predicted binding affinities than several AD therapeutics pursued in clinical trials, along with favorable pharmacokinetic properties and predicted blood–brain barrier permeability. Although experimental validation is required, these findings suggest that tau acetylation may contribute substantially to pathogenic aggregation and highlight dual-target retro-inverso peptides as a promising strategy for future AD drug development.
This article has been tagged with: