Bacteriophage TLS sensitizes Escherichia coli to antibiotics
(1) Parker High School
https://doi.org/10.59720/25-138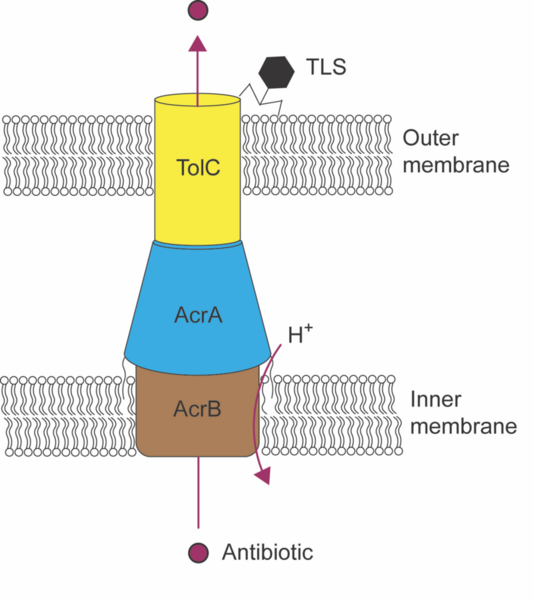
There is a significant threat to health across the world due to the misuse of antibiotics, which yield resistant pathogens. Discovering new antibiotics or using bacteriophages—viruses that infect bacteria—to treat resistant infections are two possible solutions to antibiotic resistance. A third strategy is to use phages to make current antibiotics more effective. We hypothesized that targeting the TolC protein in Escherichia coli with a bacteriophage would increase E. coli sensitivity to antibiotics. TolC is the pore-forming protein in the AcrAB-TolC efflux pump and is involved in antibiotic resistance. We used TLS, a bacteriophage that binds TolC to initiate infection, to generate TLS-resistant E. coli. We tested these phage-resistant variants for their sensitivity to the antibiotics novobiocin and chloramphenicol and performed Sanger sequencing to identify genotypic changes in the tolC gene locus that could account for changes in sensitivity. The minimal inhibitory concentration for both novobiocin and chloramphenicol was lower in all phage-resistant variants except for one, which showed sensitivity only to novobiocin. This finding indicates that E. coli variants that survive infection by TLS are sensitized to antibiotics, illustrating an evolutionary tradeoff. Interestingly, we did not identify mutations in the tolC gene that could explain the phenotypic changes aside from a point mutation in the PhoP binding site in one variant. We hypothesize that resistance to novobiocin and chloramphenicol arises not from genetic changes to tolC, but from alterations in its level of expression. Alternatively, mutations that affect lipopolysaccharide synthesis could explain the phenotypes we observed.
This article has been tagged with: