The effect of natural phenolic compounds on reducing oxidative stress
(1) Mills E. Godwin High School, (2) Department of Chemistry, University of Richmond
https://doi.org/10.59720/24-383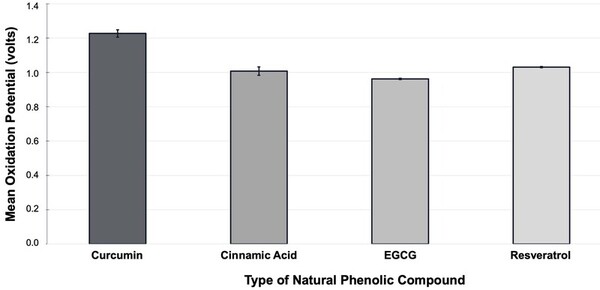
Oxidative stress is caused by oxidation-reduction dysregulation between free radicals and antioxidants, damaging cellular components. Research implicates this phenomenon in the pathophysiology of various diseases, notably cancer and neurodegenerative conditions, which are becoming more prevalent due to environmental pollutants and unhealthy lifestyle choices. Therefore, we aimed to identify a natural phenolic compound exhibiting potency in reducing oxidative stress. By investigating the antioxidant capacities of these phenolic compounds, this research contributes to our understanding of natural antioxidants and their potential applications in preventing and managing oxidative stress-mediated diseases. We used cyclic voltammetry, an electrochemical technique, to measure the oxidation potential of different compounds. We hypothesized that curcumin would exhibit the smallest oxidation potential compared to cinnamic acid, epigallocatechin gallate (EGCG), and resveratrol, due to its extensive conjugation system and electron-donating functional groups that facilitate electron delocalization. We found that EGCG was the easiest to oxidize and most effective in alleviating oxidative stress, followed in decreasing effectiveness by cinnamic acid, resveratrol, and curcumin. Our findings did not support our hypothesis, as curcumin exhibited the highest oxidation potential among the tested compounds. This contrasts with the predicted outcome based on its structural features. The results highlight the importance of molecular structure in determining the antioxidant effectiveness and suggest that EGCG may be a more promising candidate for reducing oxidative stress than other commonly studied phenolic compounds.
This article has been tagged with: