CDK7 inhibition disrupts androgen signaling and induces metabolic rewiring in prostate cancer cells
(1) Scarsdale High School, (2) University of Pennsylvania, Perelman School of Medicine
https://doi.org/10.59720/24-324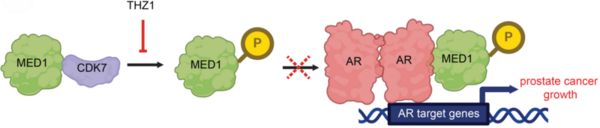
Prostate cancer (PC) is the second leading cause of cancer-related deaths among men in the United States. The lethality of advanced castration-resistant prostate cancer (CRPC), which is resistant to conventional androgen deprivation therapy, highlights the urgent need for new therapeutic strategies. In this study, we focused on how cyclin-dependent kinase 7 (CDK7) regulates transcriptional programs in PC. We hypothesized that inhibiting CDK7 would reprogram transcriptional networks in CRPC cells that may be associated with adaptive responses to treatment. To test this, we examined androgen receptor (AR) signaling pathways, which play major roles in PC proliferation. CDK7 phosphorylates Mediator complex subunit 1 (MED1), a coactivator of AR, enhancing the transcription of target genes that drive tumor growth. We assessed the effects of THZ1, a selective CDK7 inhibitor, on CRPC cell lines, including lymph node carcinoma of the prostate (LNCaP) and vertebral cancer of the prostate (VCaP). Through analysis of RNA-seq datasets, we found significant transcriptional changes following CDK7 inhibition, including downregulation of oncogenic pathways such as AR signaling and MYC targets, and notable upregulation of oxidative phosphorylation (OXPHOS). This suggests a metabolic shift that may help CRPC cells adapt to CDK7 inhibition. Gene set enrichment analysis highlighted the impact of CDK7 inhibition on metabolic and transcriptional pathways, underscoring the potential of targeting CDK7 in AR-driven PCs, including those resistant to androgen deprivation therapy. Combining CDK7 inhibitors with agents that target OXPHOS could improve therapeutic efficacy by simultaneously disrupting transcriptional and metabolic adaptations in treatment-resistant PC.
This article has been tagged with: