Effects of microconvection on bubble displacement during water electrolysis under microgravity
(1) Valley Christian High School, San Jose, California
https://doi.org/10.59720/22-246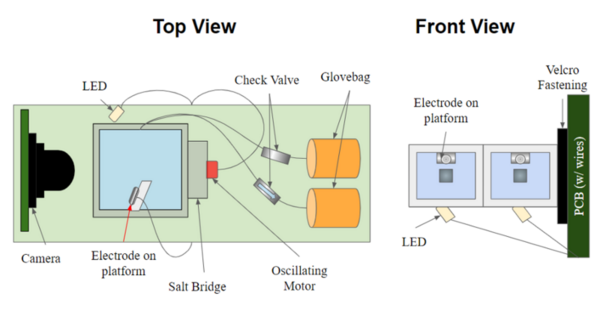
Water electrolysis is the electrochemical process of passing an electrical current through water to separate it into hydrogen and oxygen gases at two electrodes. The hydrogen and oxygen gas bubbles produced during water electrolysis are displaced from the electrodes by two classes of phenomena, which we categorize as either buoyancy or microconvection. Buoyancy is the phenomenon caused by the upward force exerted by a fluid. We define microconvection as the effects of non-buoyant bubble-displacing phenomena, such as Brownian motion. We determined the role of microconvection during water electrolysis by comparing the formation and behavior of gas bubbles under both terrestrial gravity (1G) on Earth and microgravity (μG) aboard the International Space Station. During water electrolysis under 1G, buoyancy-induced macroconvection dominates microconvection attributed to non-buoyant forces. The accumulation of gas bubbles can impede the contact of the buffer solution and platinum electrode, hindering the current flow for further electrolysis. We studied this phenomenon with a camera and current sensor. We hypothesized that microconvection has a weaker effect in displacing bubbles compared to buoyancy and that the current will decrease more over time under μG due to extended contact of the gas bubbles with the electrodes during electrolysis. Our results showed that microconvection plays a non-negligible role in bubble displacement in water electrolysis, a finding which has broad applications, including zero-pollution hydrogen production and hydrogen fuel cells.
This article has been tagged with: