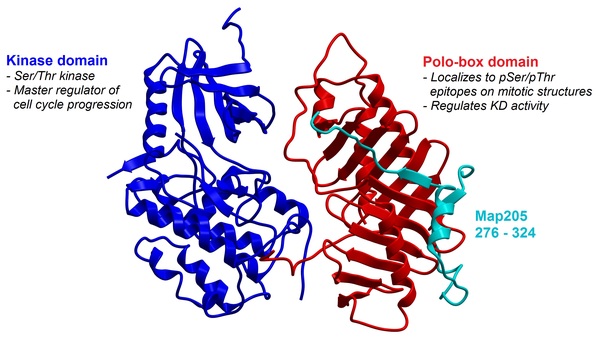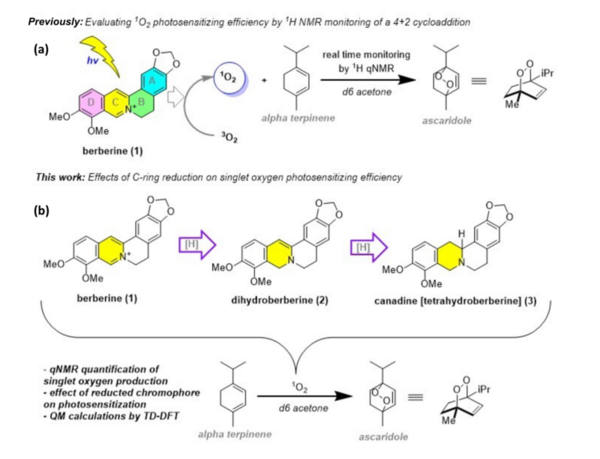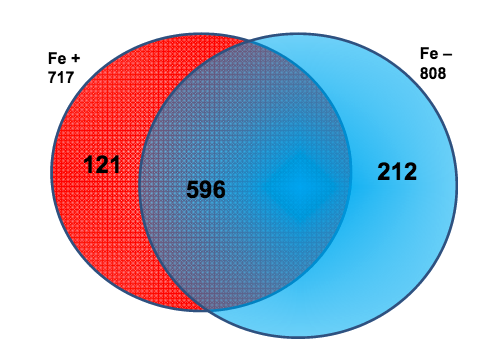
In many areas of the world’s oceans, diatoms such as Thalassiosira pseudonana are limited in growth by the availability of iron (Fe), which is an essential nutrient for diatoms. The authors of this study examined if Fe-limitation makes a significant difference in the proteins expressed within the chloroplast, the power source for diatoms, utilizing a new plastid isolation technique specific to diatoms and completing 14 mass spectrometry experiments.
Read More...