Computational development of aryl sulfone compounds as potential NNRTIs
(1) Irvington High School, Fremont, California
https://doi.org/10.59720/21-226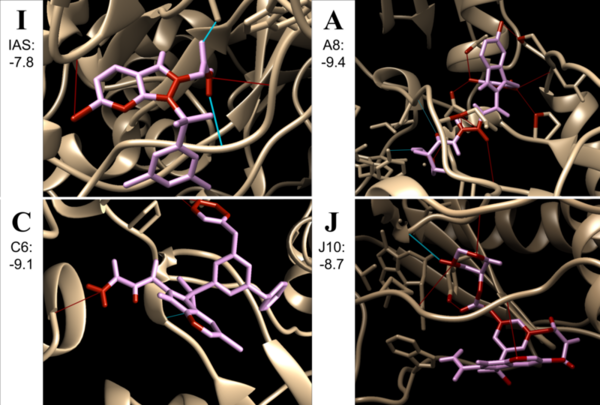
Human immunodeficiency virus (HIV) attacks the host’s immune system and affects millions of people globally. Due to the high mutation rate of HIV, it is of great importance that novel drugs are found to combat new strains. Non-nucleoside reverse transcriptase inhibitors (NNRTIs) are allosteric inhibitors that bind to the HIV reverse transcriptase and prevent replication. Indolyl aryl sulfones (IAS) and IAS derivatives have been found to be highly effective against mutant strains of HIV-1 reverse transcriptase. Here, we analyzed molecules designed using aryl sulfone scaffolds paired to cyclic compounds (that have previously been found to be beneficial groups for NNRTIs) as potential NNRTIs through the computational design and docking of 100 novel NNRTI candidates. Moreover, we explored the specific combinations of functional groups and aryl sulfones that resulted in the NNRTI candidates with the strongest binding affinity while testing all compounds for carcinogenicity. We hypothesized that the combination of an IAS scaffold and pyrimidine would produce the compounds with the best binding affinity. Our hypothesis was correct as the series of molecules with an IAS scaffold and pyrimidine exhibited the best average binding affinity. Additionally, this study found 32 molecules designed in this procedure with higher or equal binding affinities to the previously successful IAS derivative 5-bromo-3-[(3,5-dimethylphenyl)sulfonyl]indole-2-carboxyamide when docked to HIV-1 reverse transcriptase. These findings contribute to the search for novel NNRTIs as well as expanding knowledge regarding the characteristics of the NNRTI pharmacophore.
This article has been tagged with: