Developing a Portable, Reusable, and Inexpensive Magnesium-Air Fuel Cell
(1) Saint Francis High School, Mountain View, California, (2) Schmahl Science Workshops, San Jose, California
https://doi.org/10.59720/18-052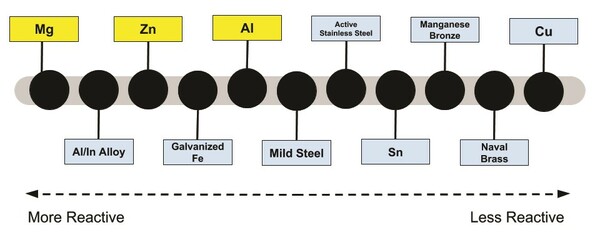
Global warming is becoming an increasingly bigger problem, and it is important that we work to find alternative sources of electricity. The goal of this project was to determine what metal is best for use in a portable and reusable metal-air fuel cell. The three metals tested for use in a metal-air fuel cell were aluminum, zinc, and magnesium. These metals were selected due to their high theoretical voltage, faradic capacity, and widespread usage as sacrificial anodes in the naval shipbuilding industry. We created a design for a portable and reusable fuel cell frame in Google Sketchup and then 3D-printed. For each of the three types of metal, we tested the fuel cell three times over a total span of six hours. Every hour, we recorded the voltage and current produced by the fuel cell. After the three trials, we averaged the data at every measurement point. Our data shows that magnesium produced the greatest voltage and current. On average, magnesium produced 197% more voltage and 740% more current than zinc. Additionally, magnesium produced a voltage 280% greater and a current 1593% greater than aluminum during the six hours of testing. This project indicates that magnesium produces the greatest voltage and current in a metal-air fuel cell, as well as the viability of magnesium-air fuel cells. Due to their long shelf life and self-sufficiency, they can be used by emergency response teams and in developing areas.
This article has been tagged with: