The peroxidase-like activity of papain colorimetrically detects H2O2 and glucose with high sensitivity
(1) Daegu International School, Dong-gu, Daegu, Republic of Korea, (2) epartment Biological Science, University of Suwon, Wau-ri, Bongdam-eup, Hwaseong, Gyeonggi-do, Republic of Korea
https://doi.org/10.59720/22-152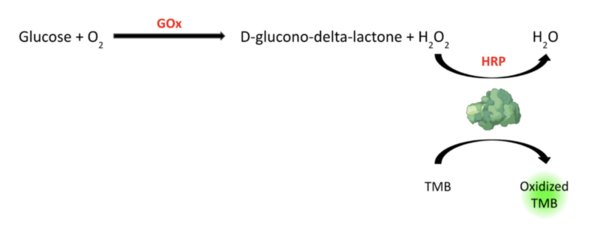
To date, several glucometer methods are available for patient-guided glycemic control, which diminishes the onset and severity of diabetic complications. In a dipstick urine glucose test, horseradish peroxidase (HRP) oxidizes tetramethylbenzidine (TMB) in the presence of hydrogen peroxide (H2O2) to quantitatively produce a colorimetric signal and predict glucose concentration. Papain, a sulfhydryl protease secreted by the latex of Carica papaya, displays HRP-like activity with high selectivity. However, the current papain-based glucometer method lacks sensitivity. We hypothesized that catalyzing the oxidation of papain under varying buffer conditions (e.g., papain, TMB, and acetic acid concentrations) would improve glucose detection sensitivity. To test this, we performed glucose oxidase (GOx)-mediated glucose oxidation and papain-mediated TMB oxidation under varying sample conditions in vitro and spontaneously coupled them. The aims of this study were to validate the coupling of GOx-mediated glucose oxidation and papain-mediated TMB oxidation and identify the optimized conditions that maximize catalytic activity. We confirmed that glucose oxidase quantitatively produces H2O2 (in a glucose concentration-dependent manner), the presence of which catalyzes papain-mediated production of a colorimetric signal. When two oxidation reactions were coupled, we identified that the sensitivity of papain’s glucose detection increased by ten-fold compared to previous studies with a markedly high correlation (R2 = 0.99). In particular, substituting sodium acetate-acetic acid (HAc-NaAc) buffer with acetic acid and increasing the sample incubation time and papain concentration markedly increased the sensitivity of colorimetric signals. The optimized protocols can be integrated into emerging hydrogel technology to develop a non-invasive glucometer patch predicting blood glucose from the patient’s produced sweat.