Strain-selective in vitro and in silico structure activity relationship (SAR) of N-acyl β-lactam broad spectrum antibiotics
(1) Mission San Jose High School, Fremont, California, (2) Dougherty Valley High School, San Ramon, California, (3) Quarry Lane High School, Dublin, California, (4) Leigh High School, San Jose, California, (5) Saint Francis High School, Mountain View, California, (6) Basis Independent Silicon Valley, San Jose, California, (7) Quarry Lane High School, Dublin, California, (8) Dublin High School, Dublin, Fremont, California, (9) ⁸Dept. of Chemistry, Biochemistry, and Physical Science, Aspiring Scholars Directed Research Program, Fremont, California
https://doi.org/10.59720/21-034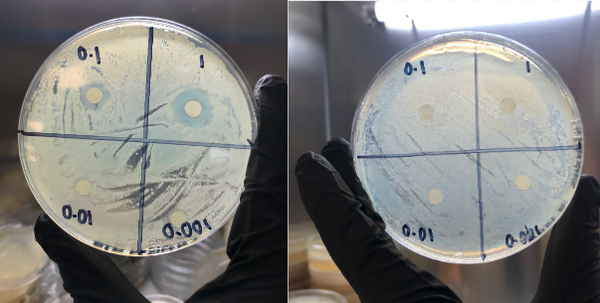
Different bacterial species have developed resistance to antibiotics over the years. Penicillin analogs have been developed to counter the bacteria’s acquired resistance to standard antibiotics; in this study, we investigate the antibacterial efficacy of penicillin G and its analogs amoxicillin, carbenicillin, piperacillin, cloxacillin, and ampicillin, against four species of bacteria. Penicillin antibiotics mimic the D-Alanyl-D-Alanine active site region of penicillin-binding proteins (PBP). PBPs are enzymes used in the synthesis of bacterial cell walls. Through a Kirby-Bauer assay, we found that the inhibition performance of these penicillin-type antibiotics varies greatly among different bacterial species. Moreover, to investigate the structural basis for the in vivo Structure Activity Relationship observed, we also conducted in silico virtual creening via homology modeling, molecular docking, and density functional theory (DFT) calculations. We concluded that all six penicillin-type antibiotics inhibit Staphylococcus epidermidis, Escherichia coli, and Neisseria sicca with varying degrees of efficacy but exhibited no inhibition against Bacillus cereus. Our computational results suggest that the distance between the β-lactam ring and the active serine is more influential than the thermodynamic binding affinity for these antibiotics to interact with the PBPs. Penicillin G had the greatest broad-spectrum antibacterial activity with a high radius of inhibition against S. epidermidis, E. coli, and N. sicca.
This article has been tagged with: